Skai Ventures: Strategies for Takeoff

TruTag Technologies: Methodically Marching to the Marketplace
In the course of researching new technologies related to the eye in the early 2000s, Wuh came across FDA-approved silica and was intrigued by the possibility that it could be optically encoded and read to create a platform to hinder counterfeiting.
Federal agencies say they regularly intercept shipments of counterfeit drugs that don’t have the proper ingredients. The Food and Drug Administration says that in some parts of the world, 30 to 50 percent of the drugs used to treat serious illnesses are counterfeit, and often contain little or none of the proper ingredients. Wuh was not the only one to see the issue as a significant threat to public health, but he was one of the few to recognize an opportunity to do something about it.
Mike O’Neill, TruTag’s chief technology officer, remembers Hank turning to him with a “science project”: Take a spectral fingerprint, encode it on a microtag that can be incorporated into a drug dosage and make it a reality. Because human applications like this require multiple approvals from international regulatory agencies, it takes years to move such a product into the marketplace, and it requires a level of patience and perseverance on the part of creators and investors not often seen in other startups.
O’Neill might have been the ideal candidate: He has a Ph.D. in biophysics from Cal-Berkeley and decades of experience in an unlikely but perfect combination of specialties – optical data storage and renewable security – plus he spent time in the submarine corps based at Pearl Harbor. He has helped to develop this leading-edge technology into a platform that recently earned TruTag recognition by the World Economic Forum as a Technology Pioneer, among other accolades.
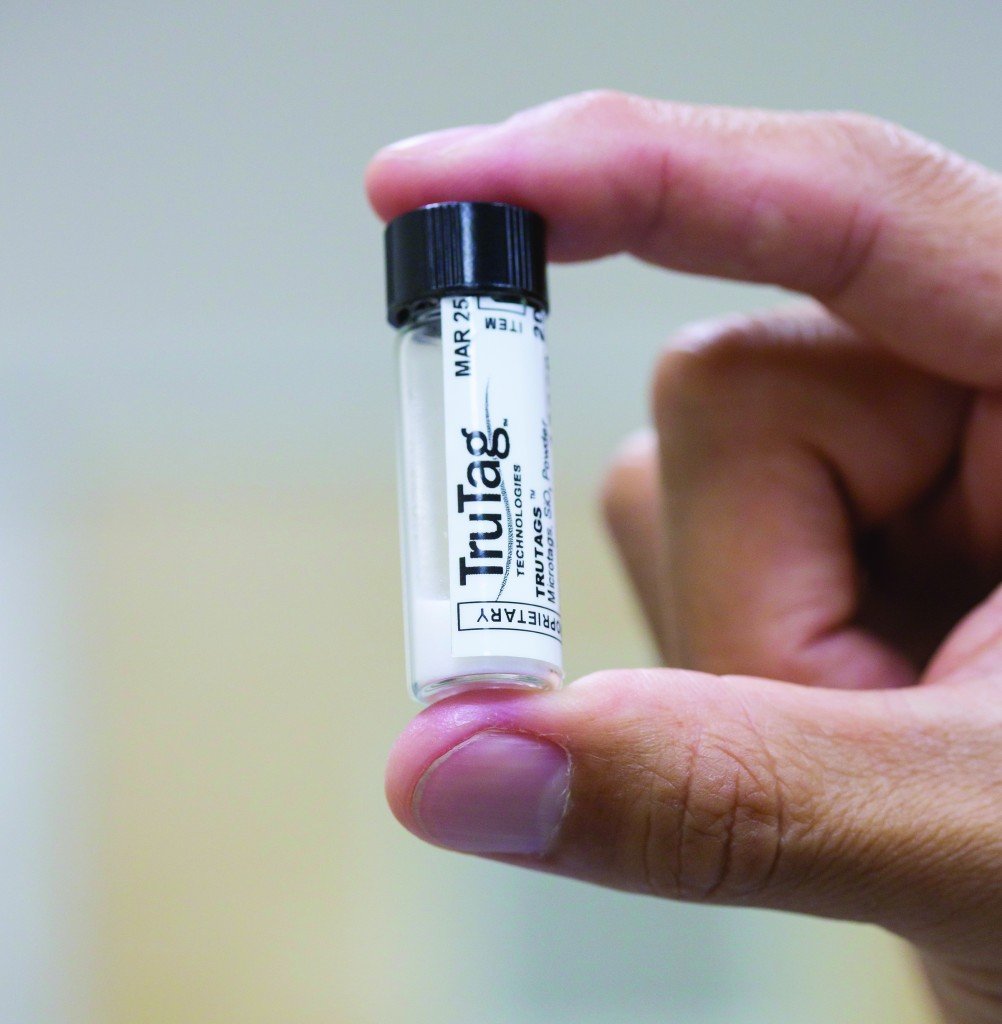
PHOTO: DAVID CROXFORD
The company’s microtags are about the size of a particle of dust and can be coated onto every pill as part of a pharmaceutical’s existing manufacturing process; they can then be scanned with a special reader to reveal vital information, such as product strength, lot number, manufacturing date, authorized customer and more. And, because microtags are encoded throughout, they still contain all of their information when broken into pieces, exposed to extreme temperatures or subjected to rough handling.
As TruTag’s “on-dose authentication” moves closer to its commercial launch, industries outside of pharmaceuticals – electronics, automotive parts and others – are starting to use the tags and readers to verify the authenticity of their products. O’Neill says he and his team are deliberately controlling the pace of delivery so the technology transfer to manufacturing and application is thoroughly vetted and uncompromising – even as the knocking at TruTag’s door from interested customers and investors gets louder.
California-based Green Project Inc. is currently licensed to apply the tags to gears they use in manufacturing recycled ink and toner cartridges; the company buys back some of its products from distributors and sends them to TruTag to forensically confirm that no one has substituted counterfeit versions. According to O’Neill, the current partnership with Green Projects is “right-sized for ensuring that all of our systems are validated,” even though TruTag can scale up for much larger industrial applications.
The tags and the readers are now in production. It’s the application of the tags that requires the greatest degree of customization, since a customer’s particular security issue and product design dictate how, where and with what material to apply the tags. That’s why TruTag is zeroing in on opportunities that leverage a limited number of applications to achieve a high volume, and looking at subscription models to reach its efficiency targets. Toward that end, investor and partner WuXi PharmaTech, listed on the New York Stock Exchange, plans to incorporate microtags into the manufacturing processes of its many pharmaceutical customers around the globe.
TruTag’s carefully stepped approach includes ramping up manufacturing capacity in the pilot facility and ultimately building more plants as the market scales. For now, the Kapolei plant – which conforms to Good Manufacturing Practices required for pharmaceutical and food products – is “cleaner” than it needs to be for non-GMP-regulated manufacturing. That seems fine with TruTag’s scientists and entrepreneurs, who think the company is poised to become the overnight success it has been preparing for these last six years.








